Answer
Chronic/late RV perforation
Explanation
Another month later the patient presented to A&E with chest pains and a sensation that the pacemaker was delivering shocks. The CT report from the previous week was chased and revealed the following.
“Dual-chamber cardiac pacemaker is noted in situ. The tip of the right ventricular lead is seen located beyond the confines of the myocardium and abutting the pericardium. Features suggest RV lead perforation of the myocardium, on the background of clinical history provided. Urgent specialist Cardiac review is recommended.
The lead is seen traversing through the pericardial space. However, the pericardium appears grossly intact. No evidence of pericardial effusion”
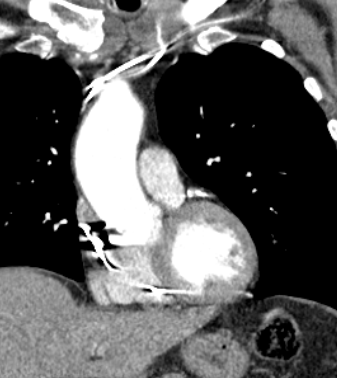
Figure 3 – CT scan image showing the RV lead tip beyond the myocardial border
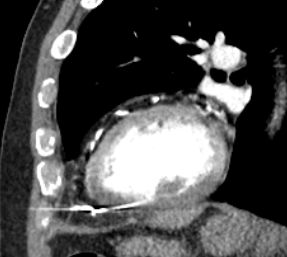
Figure 4 – CT scan image showing the RV lead tip within the pericardial space
The patient was admitted to hospital to wait for transfer to a tertiary centre for system extraction. A further pacing check was performed which found the R wave had deteriorated to the point where the device was no longer able to sense. Unfortunately, the patient contracted Covid-19 whilst in hospital so transfer was delayed. We are yet to hear the outcome of this case from the tertiary centre.
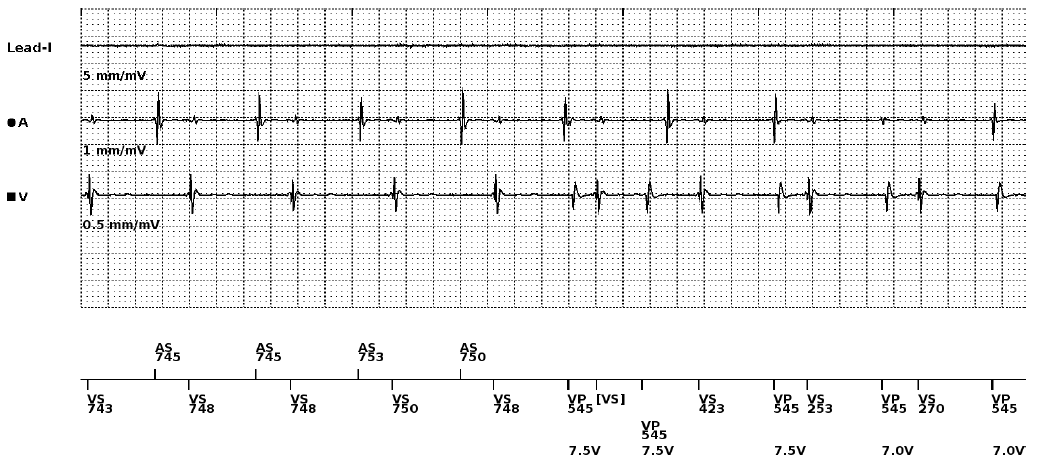
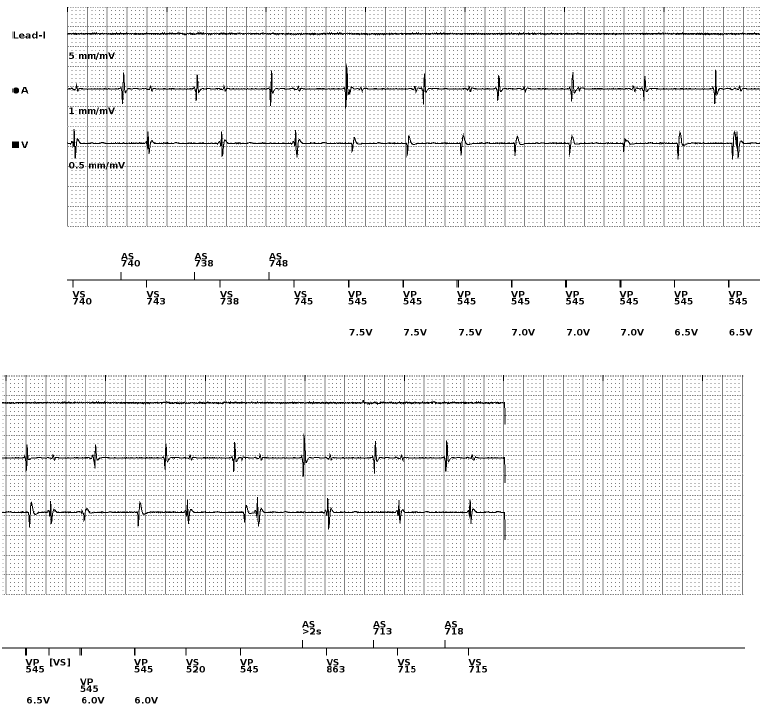
Due to the original finding that the unipolar threshold was higher than the bipolar threshold there was a suspicion of perforation, however as the lead impedance was still in range, the patient was asymptomatic and there was no effusion on echo, the level of suspicion was not very high, so the CT was not requested as an emergency. In hindsight this impedance trend is likely due to an incomplete or microperforation of the lead tip, or because there was no pericardial effusion present which would cause a classically low impedance outside of normal range. There may be a combination of both these explanations before and after the threshold rise. The RV lead impedance trend graph shows a sudden drop in impedance post implant but the pacing threshold was 1V @ 0.4ms in clinic at 6 weeks. Impedance and R wave trends continued to decline very gradually from that point onwards.
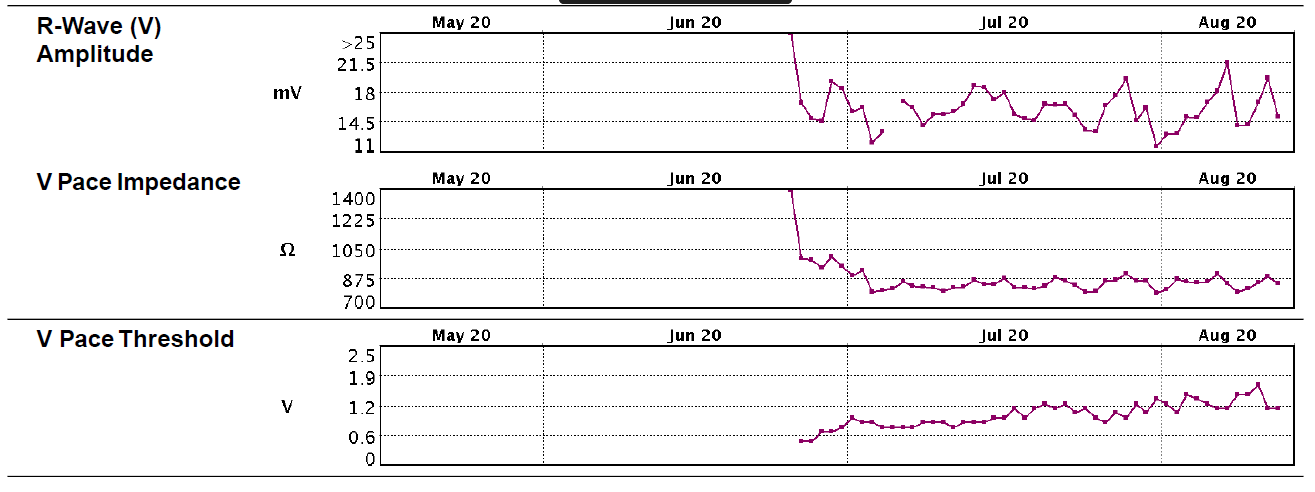
Figure 5 – RV lead trend graphs downloaded at the 6 week check
We have also seen another patient the same week who again presented 2 years post implant, but with pericardial effusion which required draining. The post procedure CT showed an RV lead tip perforation and the device was subsequently checked. There was no pacing at max output and no sensing. This patient also exhibited a significant impedance drop from 1600 Ohms to 650 Ohms (also still within normal range) with an increase in threshold at 6 weeks to 2V @ 1ms which was assumed to be due to microdisplacement due to CXR findings. Unfortunately, the unipolar threshold value was not documented but noted to be “no better than bipolar”.
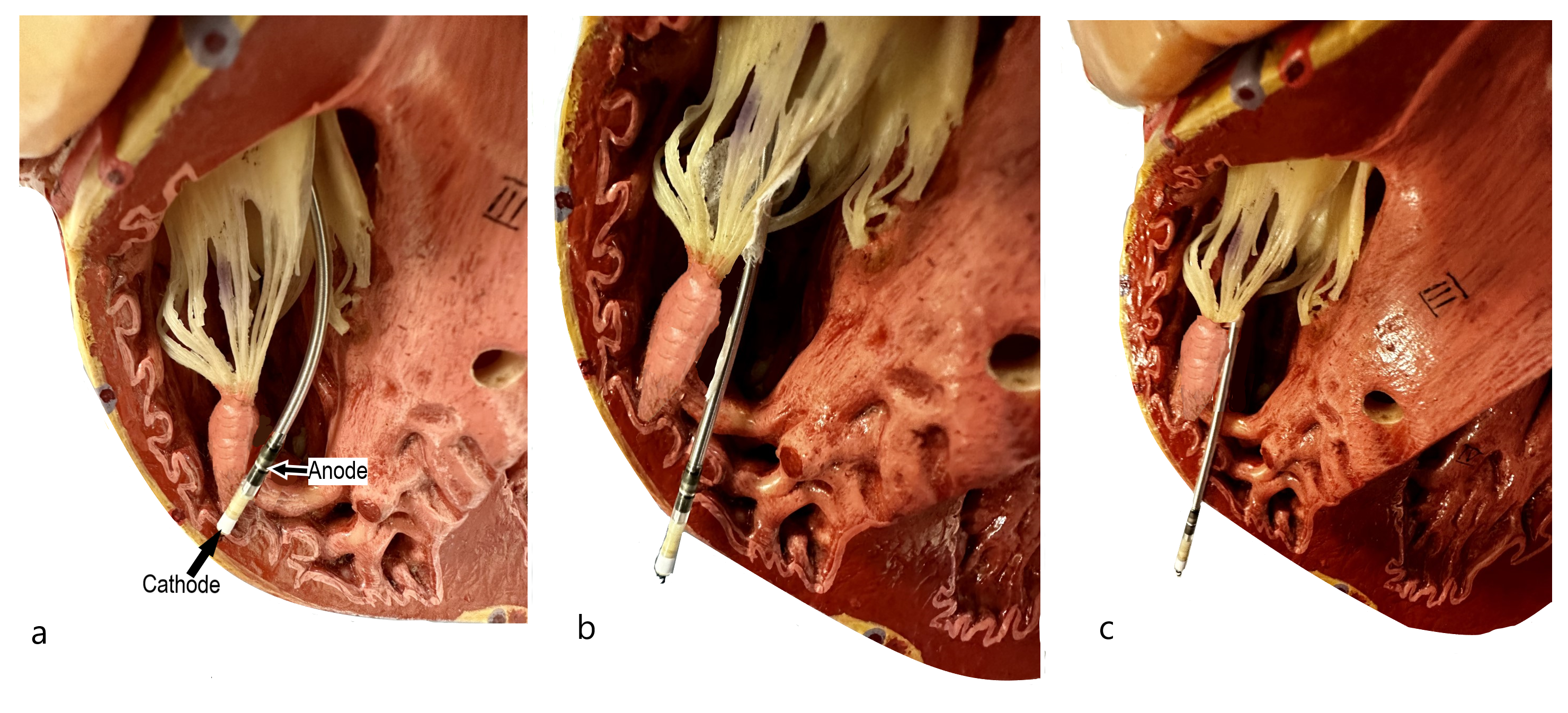
Figure 6a – Active fixation lead where cathode is within RV free wall and anode within RV cavity. 6b- Cathode has entered the pericardial space but anode is within the myocardium. 6c- Both cathode and anode are now within the pericardial space.
In bipolar pacing the stimulus moves from the device to the lead tip (cathode) and then returns to the ring electrode (anode). In unipolar pacing the anode is the device itself and the stimulus returns via the body tissues. If the cathode is surrounded by pericardial effusion this lowers the unipolar impedance significantly as it is not in contact with myocardial tissue and also significantly increases the output as the circuit is extracardiac and not able to directly stimulate the myocardium. If the bipolar anode is still within the myocardium (figure 6b) the bipolar threshold remains lower but outside normal range as the heart is still able to be stimulated at high outputs when the impulse returns to the ring electrode. If the bipolar ring also passes beyond the myocardium (figure 6c) then loss of bipolar capture and sensing can also occur.
Discussion
It is not clear how many cases there are of perforation in asymptomatic patients or those who do not present with pericardial effusion due to low suspicion or assumption of chronic lead deterioration. However Sidhu, Ranjani and Rinaldi (2019) have presented a case where an asymptomatic patient underwent new RV lead implant due to an increased threshold of 1.75V @ 1ms. The patient was then investigated for perforation of the new lead due to an onset of symptoms. It was assumed that the new RV lead had perforated the RV however it was discovered on CT that the 14 year old chronic lead was the culprit which lead to the patient needing a further procedure to extract the lead 2 months later, increasing the risk of complication in comparison to it having been extracted during the lead replacement procedure.
Ellenbogen (2014) suggests that microperforation may be suspected if the unipolar tip pacing threshold is significantly higher than the ring threshold as we found with our patient. They also advise to be suspicious of patients presenting with pectoral muscle stimulation.
RV lead perforation is mostly expected to be an acute complication leading to pericardial effusion and cardiac tamponade, however there is evidence that some patients are presenting with chronic perforation or microperforation who may exhibit atypical symptoms and lead measurements. If there is a rise in bipolar RV threshold post implant (especially if accompanied by any significant drop in pacing impedance) then unipolar threshold should be tested and documented. Patients with a higher unipolar threshold can then be highlighted to their consultant cardiologist and investigated for suspected lead perforation in the early stages, and potentially avoid a late presentation with patient compromise.
References
Sidhu, B., Rajani, R., and Rinaldi, A. (2019) ‘Chronic ventricular lead perforation: Expect the unexpected.’ Clinical Case Reports. 2019 Mar; 7(3): 465–468. Available at: https://europepmc.org/article/PMC/PMC6406153 (Accessed: 13/12/2022)
Ellenbogen, K. (2014) ‘Cardiac Pacing and ICDs’ 6th Ed. Oxford: Wiley Blackwell