Answer
Autocapture adaptive pacing was switched off. There was a consistently normal RV threshold (0.5V@0.4ms). Inappropriate capture management was noted, and the impression was of inappropriate evoked response due to hyperpolarization.
Lead revision was cancelled, and the patient was well at the 3-month follow-up appointment.
Discussion
- Ventricular capture management is an automatic pacing threshold adjustment algorithm that automatically measures pacing threshold through detection of the evoked response after a pacing stimulus.
- This algorithm then automatically updates the pacing output to optimal value. This function avoids unnecessary high output pacing and is considered to prolong battery longevity.
- Pacing threshold search is initiated according to the schedule and frequency programmed by the clinician in pacemakers (nominally set to ‘day at rest’).
- After implant detection complete: the device initiates the first pacing threshold search 12 hours after.
- If a search cannot be completed, the device retries after 30 minutes.
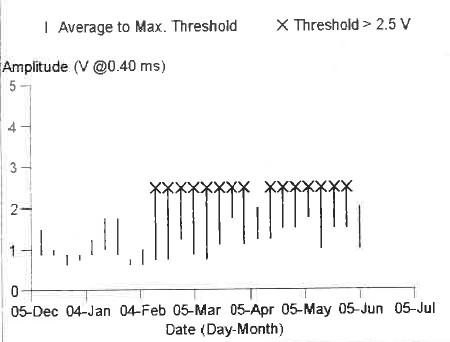
- A ‘High Threshold Condition’ warning is issued if the amplitude threshold is greater than 2.5V. The pacemaker responds by adapting to an amplitude of 5.0V and pulse width of 1.0ms. It will not give you an exact RV threshold measurement (Figure 1).
Figure 5: Ventricular capture management
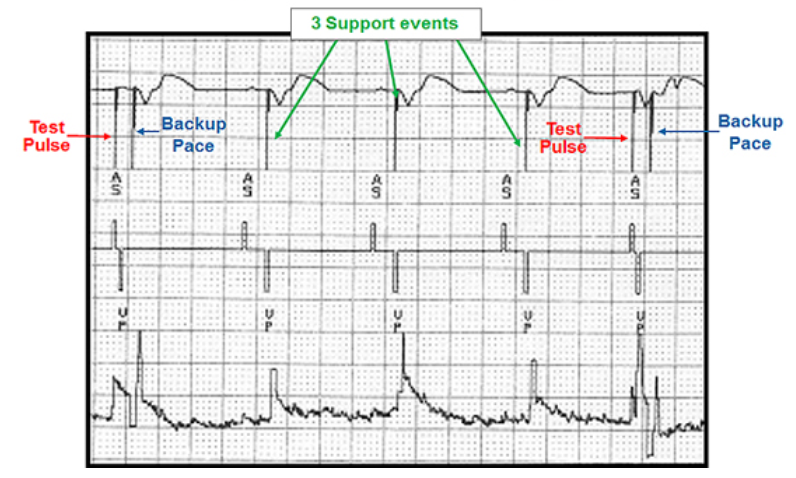
- Figure 5 shows ventricular capture management with a pacing stimulus or ‘test pulse’ being delivered to the right ventricular. When there is no evoked response detected during the pacing search within a timed interval, back-up pulse is delivered, followed by 3 further pacing pulses or ‘support events’.
- Capture verification efficiency relies on measurement of the evoked response and works by triggering a blanking window (closed) followed by an open window.
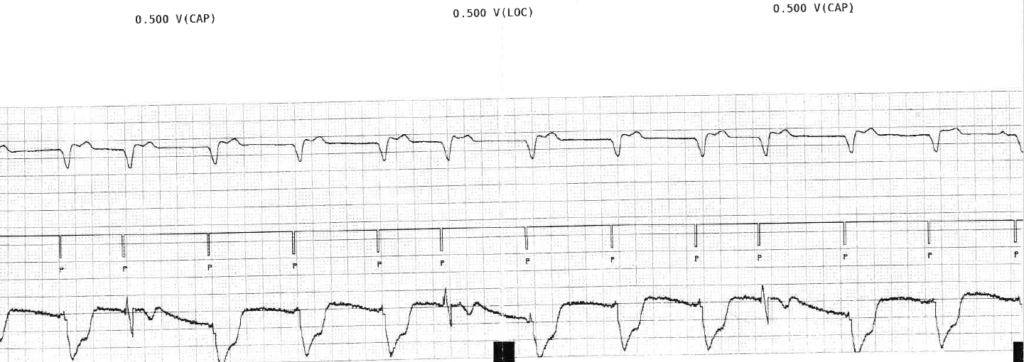
- If the device detects an ER within this second window, capture is confirmed (Figure 6). Otherwise it will deliver a 5V back-up pulse to ensure capture (Figure 7).
Figure 6: Capture verification
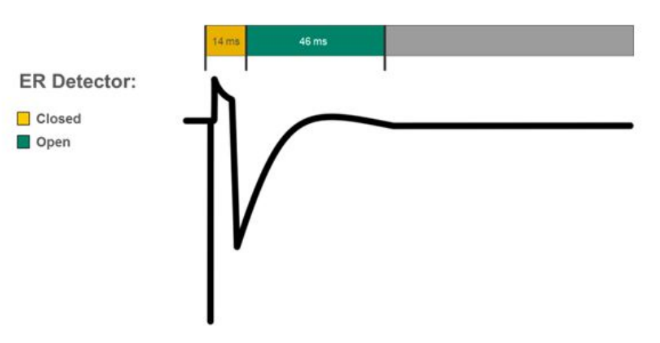
Figure 7: Capture verification with back-up pulse
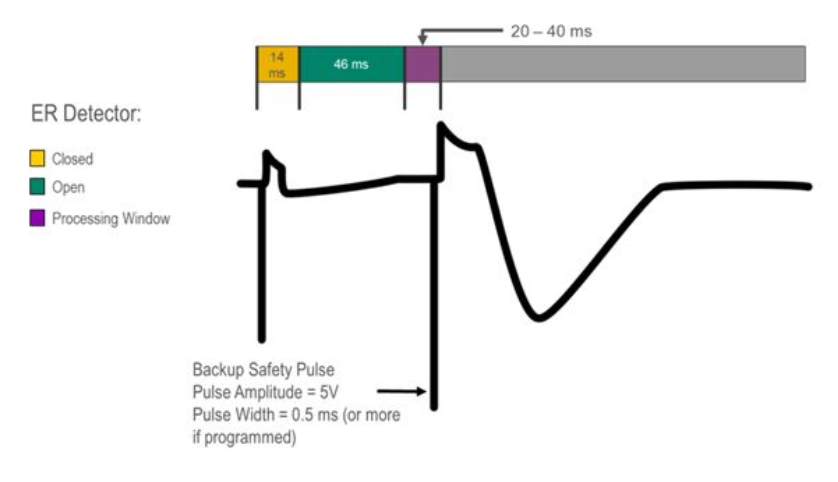
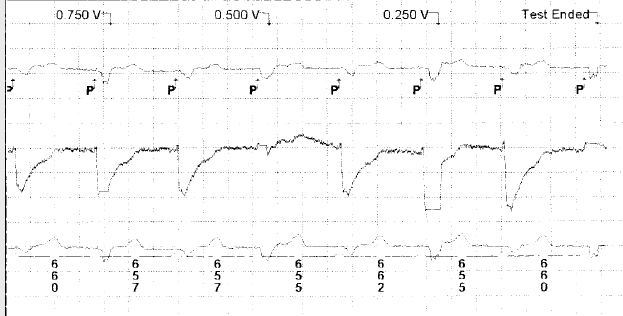
Figure 8: Ventricular capture management in our patient
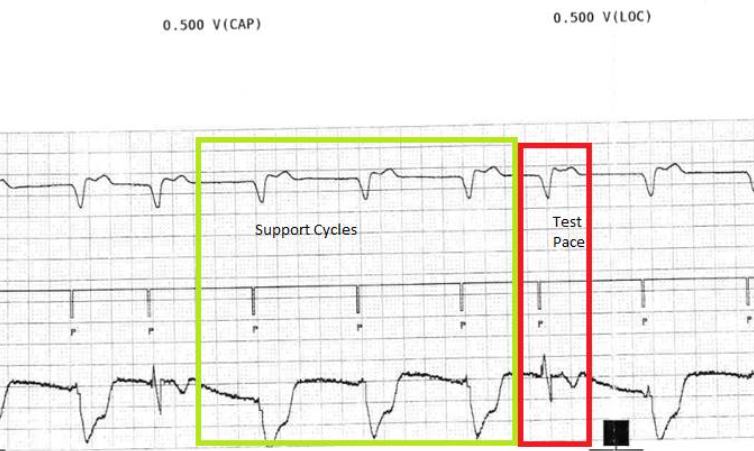
- Figure 8 shows that the device is allowing 3 support cycles as programmed rate and output followed by a faster test pace. The test pace automatically has a backup pace at programmed voltage at 1.0ms that occurs 110msec after the test pace. These backup pulses are not seen on this strip.
- This cycle repeats looking for the 2/3 capture before moving on or 3/3 capture to confirm measurement.
- Looking at the timing of the rhythm then it seems that all test paces capture as the ventricular event does not shift 110msec later. However, the evoked response signal is not consistently detected by the device so that it sometimes labels this as LOC.
- Evoked response detection and limitations: In rare instances, the pacemaker may not detect the electronic waveform created by the contracting myocardium immediately following a pacing pulse.
- High thresholds may be appropriate and due to lead dislodgement, or incomplete connection of the lead in the header block.
- Acute effects of lead maturation can also cause failure to detect the evoked response. Hyperpolarization of the EGM leading to signal saturation or changes in lead tip and muscle interface (i.e. tissue fibrosis) may be responsible for this.
- In our patient, the phenomenon was seen following the lead maturation period (typically within 4-12weeks) when fibrosis between tip to tissue interface is expected. The inflammatory reaction and the resulting fibrous tissue that occurs after implantation may act as an insulating shield around the electrode, effectively raising the threshold for stimulation.
- Remote transmission has increased during the COVID 19 pandemic. This case highlights the importance of inpatient/face-to-face review and interpretation of automated diagnostics.
With acknowledgements to Dr Greg Mellor, Consultant Cardiologist and Electrophysiologist, Royal Papworth NHS Trust
References
-
Antretter H, Bonatti J, Cottogni M, et al. Eine neue Herzschrittmacher-Stimulationstechnologie (Autocapture) ermöglicht bei unveränderter Lebensdauer die Reduktion der Generatorgrösse auf die Hälfte [A new cardiac pacemaker stimulation technology (autocapture) allows, with unchanged life expectancy, reduction of generator size by half]. Acta Med Austriaca. 1996;23(5):159-164.
-
https://www.medtronicacademy.com/features/ventricular-capture-management-feature
-
Kishihara, Jun & Niwano, Shinichi & Fukaya, Hidehira & Nishinarita, Ryo & Horiguchi, Ai & Nakamura, Hironori & Igarashi, Tazuru & Ishizue, Naruya & Fujiishi, Tamami & Yoshizawa, Tomoharu & Oikawa, Jun & Satoh, Akira & Murakami, Masami & Ako, Junya. (2017). Pacing failure caused by automatic pacing threshold adjustment system. Journal of Arrhythmia. 33. 10.1016/j.joa.2017.05.005.
-
-
HRS/EHRA/APHRS/LAHRS/ACC/AHA worldwide practice update for telehealth and arrhythmia monitoring during and after a pandemic (2020) https://doi.org/10.1016/j.hrthm.2020.06.010
-
Murgatroyd FD, Helming E, Lemke B et al. Manual vs. automatic capture management in implantable cardioverter defibrillators and cardiac resynchronization therapy defibrillators Europace (2010) 12, 811–816 doi:10.1093/europace/euq053