Answer
Undertake a further diagnostic step, or steps, with a view to device reprogramming.
Explanation:
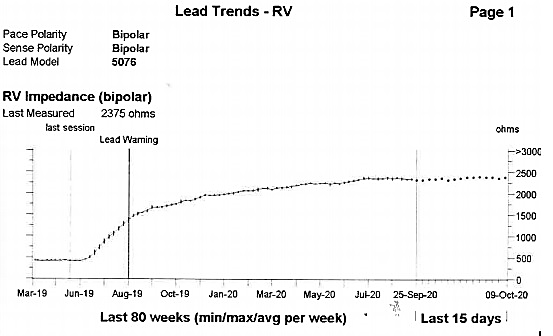
Figure 1 displays the bipolar RV lead impedance trend recorded over a period of approximately 18 months. Between late June and August 2019 there is a sharp rise in impedance from approximately 500 ohms to 1500 ohms where a lead warning is triggered. The impedance trend continues to rise gradually over time before stabilising at a range between 2250-2500 ohms, recorded at 2375 ohms at the scheduled in clinic review.
A significant rise in RV bipolar impedance as seen in this trend accompanied with the inability for the RV lead to capture at maximal outputs is highly suggestive of disruption/fracture of the RV lead outer conductor and would prompt consideration for lead replacement. In patients with a high ventricular pacing burden this is likely to require an urgent admission and would usually be discovered with a patient presenting with bradycardia related symptoms.
In this scenario the patient was asymptomatic; there was no significant RV pacing burden and good AV conduction. Consequently, review of other diagnostic information was undertaken to help plan a management strategy.
Figure 2. RV electrode impedance trend (including unipolar configuration).
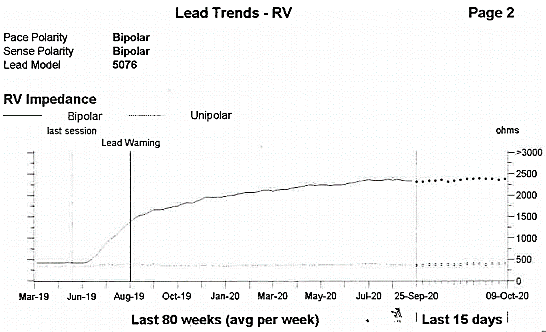
Figure 2 displays the RV lead impedance trend with both bipolar and unipolar impedance measurements. The unipolar impedance provides a measure of inner conductor integrity and shows a consistently stable impedance measurement of approximately 400 ohms, seen as a flat line which indicates that the inner conductor is intact. There was acceptable sensing, and capture thresholds in bipolar/unipolar, and unipolar configurations respectively (R-wave 14.3 mV bipolar / 11.9 mV unipolar, unipolar configuration impedance (Z) = 399 Ohms (Ω), RV threshold 2V at 1.0ms).
Acceptable measurements with a unipolar pacing configuration provided the opportunity for a conservative reprogramming strategy to be considered instead of scheduling the patient for a lead replacement which in this age group and as a second procedure comes with procedural and infection risks.
Further steps were taken to determine the safety of a programming strategy over scheduling of an invasive procedure including referral for a chest X-ray and assessment of AV nodal conduction. The chest X-ray was used to check the lead position with a view to identifying any lead defects or fractures: no macroscopic defects or fractures were seen.
A significant rise in RV bipolar impedance as seen in this trend accompanied with the inability for the RV lead to capture at maximal outputs is highly suggestive of disruption/fracture of the RV lead outer conductor¹. and would prompt consideration for lead replacement. In patients with a high ventricular pacing burden this is likely to require an urgent admission and would usually be discovered with a patient presenting with bradycardia related symptoms.
To assess the likelihood of progression to high-grade AV block requiring regular ventricular pacing, the AV nodal Wenckebach point was determined by way of high-rate pacing in the atrium to assess the longest A-A interval that fails to result in consistent 1:1 AV nodal conduction² 1:1 AV conduction was maintained at an atrial paced cycle length of 571 msec (105 ppm) and knowing this was in the presence of negative dromotropic medication, in this instance Bisoprolol and Flecainide which would will likely impact on this patient’s Wenckebach cycle length, it was felt that this was acceptable AV conduction for his age.
Of note for this age group, at rest, an ideal paced AV Wenckebach response would be 1:1 AV conduction at AAI rates ≥ 120/min (120 ppm)³ When AV conduction is significantly shorter than 100 ppm without such medication on board, infra-Hisian conduction system disease is strongly suspected; such an observation would be associated with an increased propensity to develop future high-grade AV block.3 If this had of been the case scheduling for a lead replacement may have been the preferred management plan.
As the patient was not pacing dependent in the ventricle, and moreover not reliant on regular ventricular capture, and as his unipolar ventricular lead impedance was found to be of an acceptable range, simply reprogramming the RV lead to the unipolar pace configuration obviated the need for this patient to undergo an invasive procedure at this stage.
Following this finding and device reprogramming, intensified pacing clinic follow-up was implemented to monitor the patient for bradycardia related symptoms, loss of AV nodal conduction and increases in RV pacing burden with a view to plan a lead replacement if signs of patient or lead deterioration were found.
This case of an asymptomatic elderly gentleman with sick sinus syndrome and preserved AV conduction, illustrates how a routine annual pacing check can throw up unexpected findings with potential prognostic important clinical consequences. Furthermore, it highlights the importance of reviewing all diagnostic information available and how knowledge of lead design and programming strategies can help determine whether a conservative or invasive approach is required to manage abnormal lead function findings.
References
1. Cardiac Pacemakers: Function, Troubleshooting and Management: Part 1 of a 2-Part Series. J Am Coll Cardiol 2017; 69: 189-210
2. Atrioventricular Wenckebach point and progression to atrioventricular block in sinoatrial disease. Haywood GA, Ward J, Ward DE, Camm AJ. Pacing Clin Electrophysiol 1990; 13 (12 Pt 2): 2054-8
3. AV Wenckebach point as a long-term predictor of right ventricular pacing dependence. Dussault C, Laflamme D, Roux J, Badra-Verdu M, Ayala-Paredes F. Can J Cardiol 2015; 31 (10): S258-259.